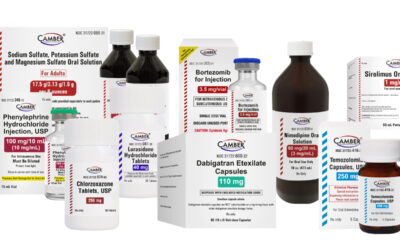
Camber Launches Sirolimus Oral Solution

Piscataway, NJ, January 29, 2025 –Camber Pharmaceuticals is excited to announce the addition of Sirolimus Oral Solution.
Sirolimus Oral Solution is an mTOR inhibitor immunosuppressant indicated for the prophylaxis of organ rejection in patients aged greater or equal to 13 years, receiving renal transplants.
- Patients at low to moderate immunologic risk: use initially with cyclosporine (CsA) and corticosteroids. CsA withdrawal is recommended 2 to 4 months after transplantation.
- Patients at high immunologic risk: use in combination with CsA and corticosteroids
Sirolimus Oral Solution 1 mg/mL is now available in a 60 mL bottle.
To find out more information on Sirolimus Oral Solution, please visit: www.camberpharma.com/sirolimus
Recent articles
- Camber Launches 13 New Generic Products in Q1 2025, Sets Ambitious Pace for the Year
- Camber Launches Mycophenolate Mofetil Capsules
- Camber Launches Sodium Sulfate, Potassium Sulfate and Magnesium Sulfate Oral Solution
- Camber’s Triple Play – Featured Generics
- Big Game Commercial
- Charting a Bold Path for 2025